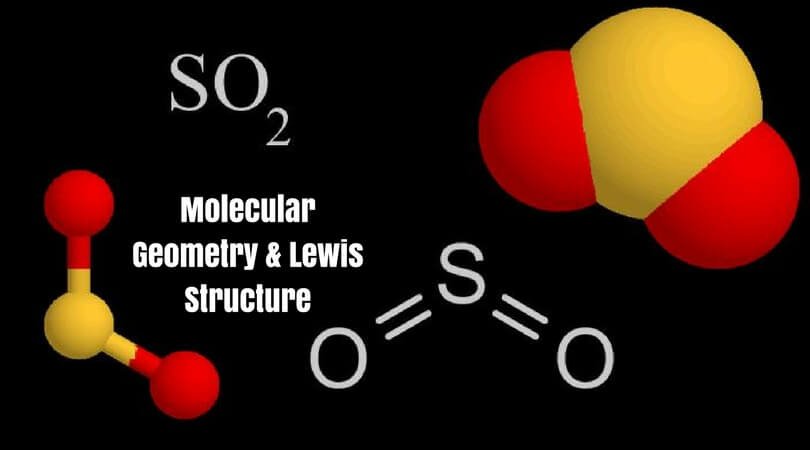
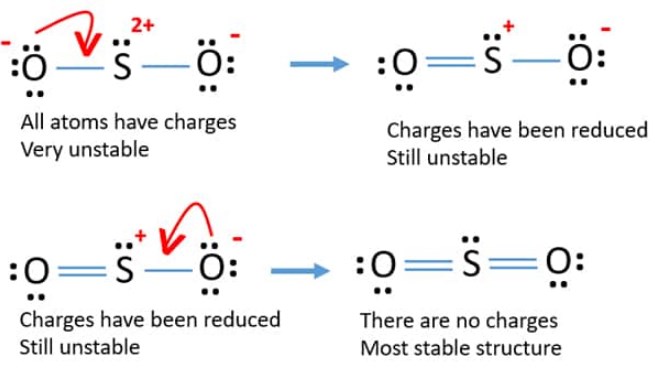
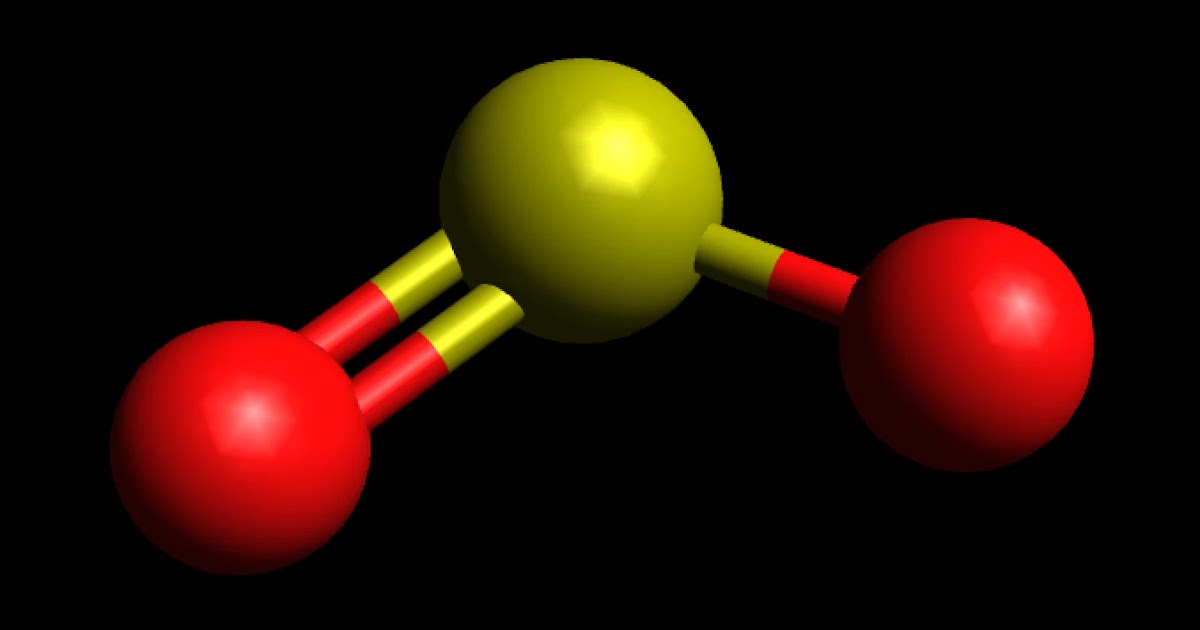
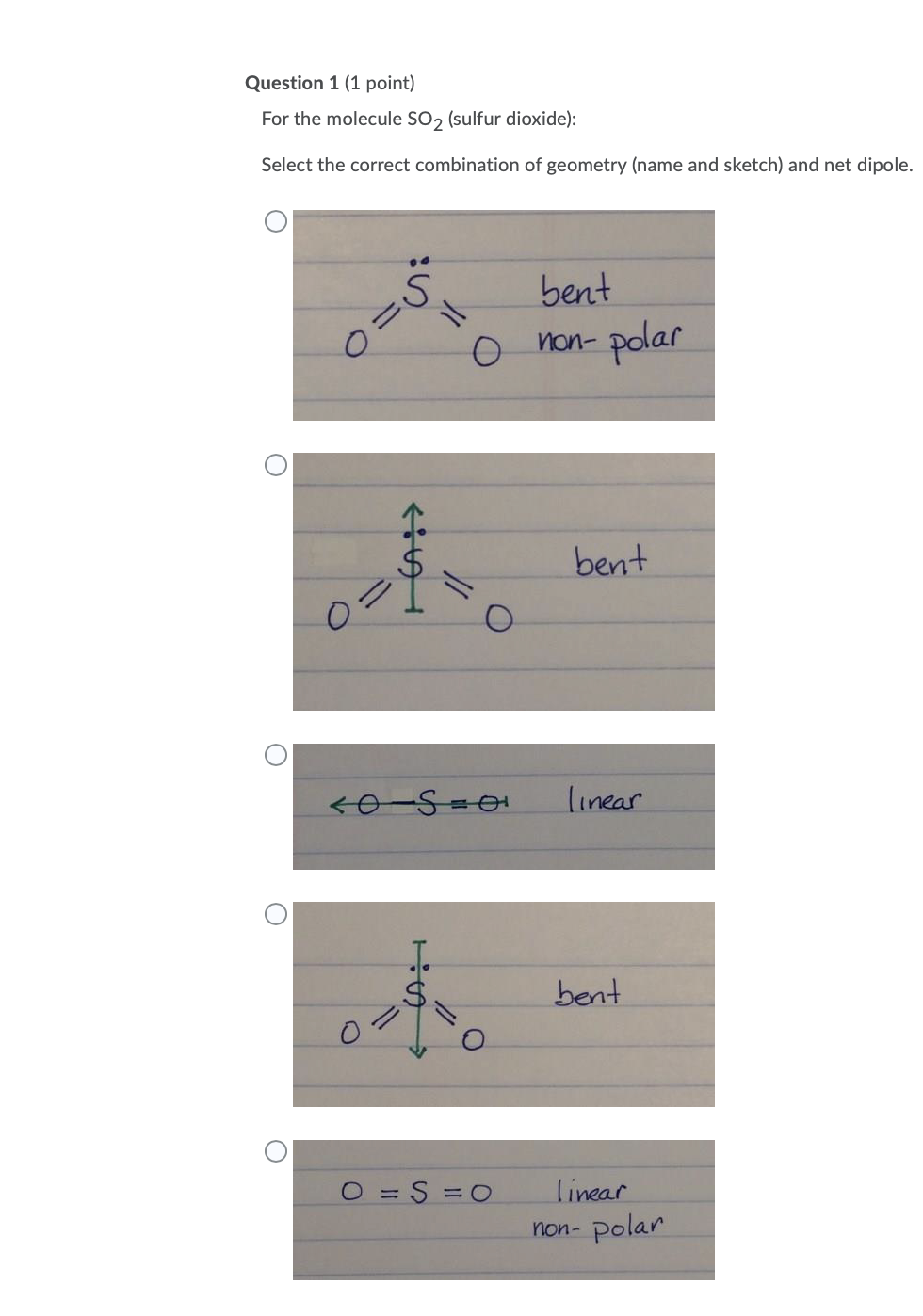
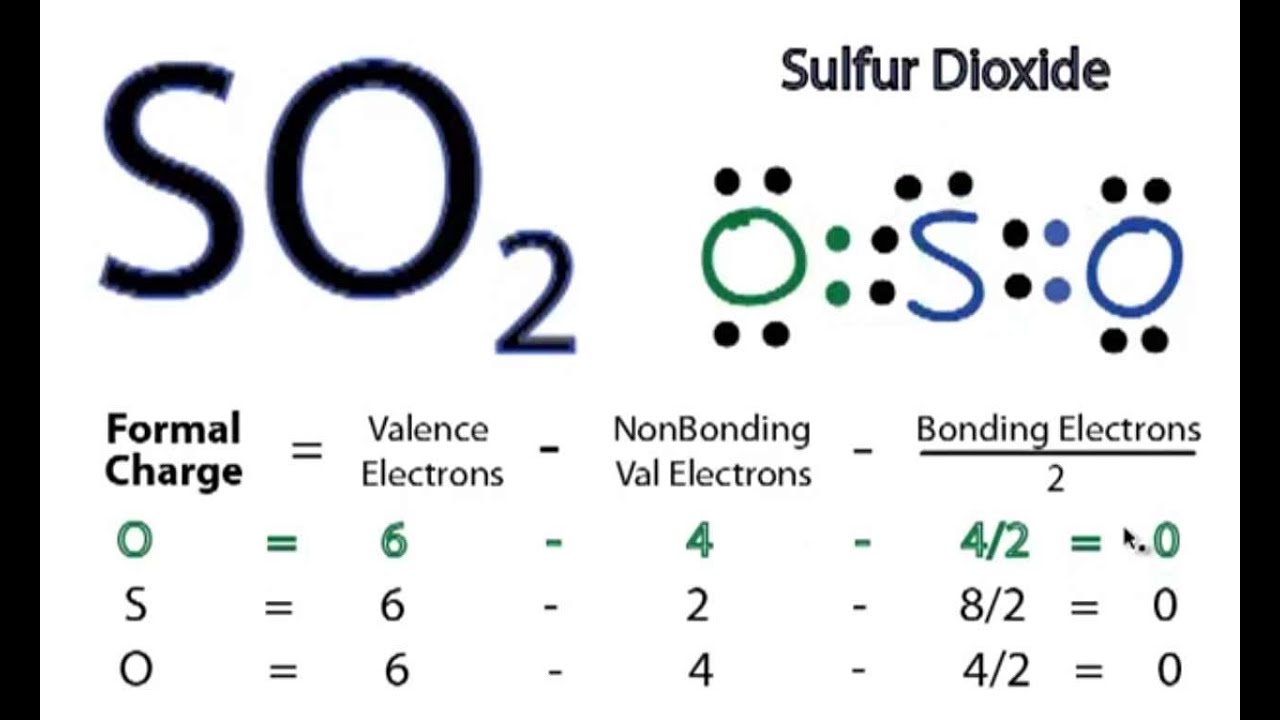
Determine the following for SO2. a. Electron geometry b. Molecular geometry (shape) c. Is the molecule symmetrical or asymmetrical? d. Is the molecule polar or nonpolar? | Study.com

CO2 and SO2 both are triatomic molecules; but there is a big difference in their dipole moment why - Chemistry - Chemical Bonding and Molecular Structure - 6914070 | Meritnation.com